
A large number of in-vitro studies have revealed intriguing pharmacodynamics of curcumin as a neuroprotective agent, specifically confirming the modulation of genes, proteins, transcription factors, and neurotransmitters at the mitochondrial level in support of curcumin’s possible positive influence on various brain health functions like neuro inflammation, neurodegenerative etc. The real challenge is delivering the free form of curcumin (unconjugated form) to the targeted brain tissues. The native form of curcumin is BBB permeable. But once it has undergone biotransformation (metabolized into glucuronides) the ability to pass the barrier is lost since the conjugated metabolites are bulky water soluble molecules and undergoes quick renal clearance.
Another point to be noted is that consumption of high dosage of unformulated curcumin 95 has shown to deliver some amount unconjugated free forms (less than 10 ng/g level) to the brain tissues when analyzed by sensitive tandem mass spectrometric tools. Thus any formulation having unformulated curcumin 95 can deliver trace amount of neuro protection, provided if we consume high dosage and the effects will be quite marginal.
Here comes the real world significance of a formulation which can deliver significant amount of free curcuminoids to the brain tissue without having the burden of high dose. CurQfen® was the solution for this dilemma.
CurQfen®, a new generation curcumin for brain health which is a green formulation of curcumin and de-bitterised fenugreek fiber as self-emulsifying hydrogel scaffold, is capable of delivering high level of BBB-permeable ‘free curcumin’ even at a low dosage of 250mg single dose (Kumar et al., 2016).
CurQfen delivers >70% of the absorbed curcuminoids in the bioactive FREE form
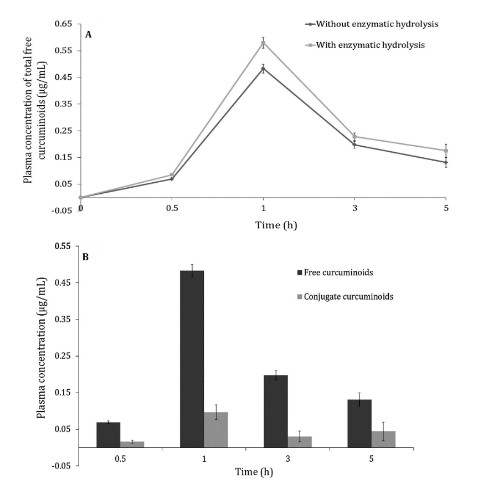
(A) Plasma concentrations of free curcuminoids by time plot following the oral administration of single dose of 1000 mg CGM containing 391 mg of total curcuminoids. Plasma concentrations were calculated with and without enzymatic hydrolysis with β-glucuronidase enzyme. Data expressed as mean ± SEM, where p < 0.05, when the plasma free curcuminoids concentrations with and without enzymatic hydrolysis was compared. (B) The relative distribution of free curcuminoids and conjugated metabolites at different post-administration time interval (Kumar et al., 2016).
Improved BBB-permeability and Brain pharmacokinetics of CurQfen: The litmus test for Brain bioavailability
CurQfen® has shown to deposit free curcuminoids, (including curcumin, DMC & BDMC) at >100 ng/g in brain tissues, with extended elimination half-life of 4 hours. Standard unformulated curcumin, on the other hand, detected only 1.4 ± 0.8 ng/g of curcumin in the brain tissues, indicating the improved BBB permeability.
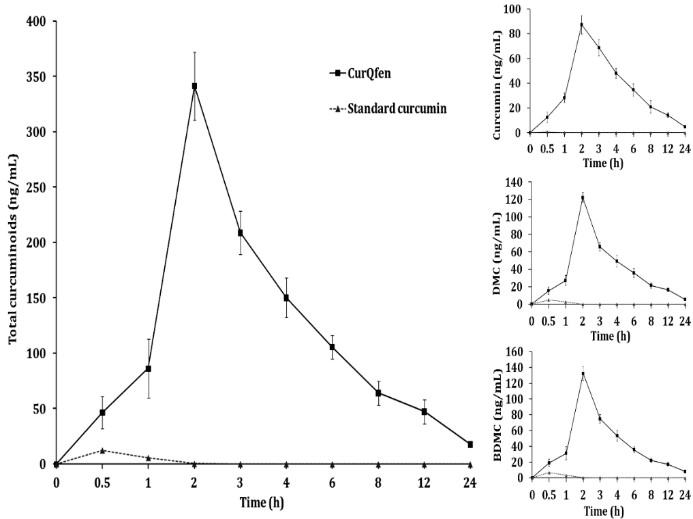
Brain concentration – time curve of total free curcuminoids and individual curcuminoids (curcumin, DMC and BDMC) of Wistar rats orally administered with standard curcumin or CurQfen. The values are presented as mean ± SD (Krishnakumar et al., 2015).
CurQfen® delivers free curcuminoids in the Hippocampus. The first line of molecular evidence for its Cognitive support
Most recently published clinical trial on Nutritional Neuroscience, 2021 revealed the brain regional pharmacokinetics following the oral consumption of CurQfen® and its effect on cognitive health.
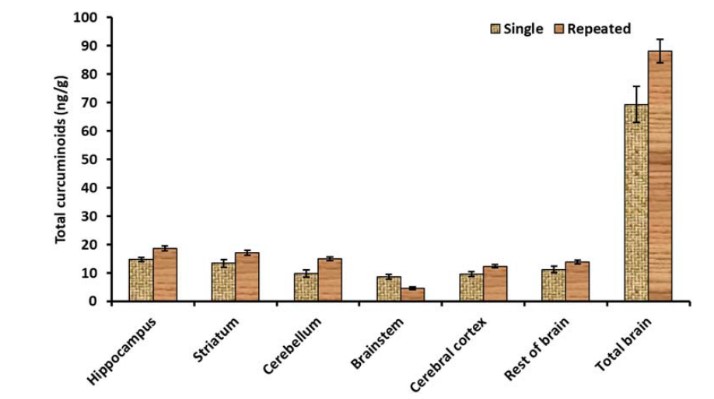
Relative distribution of curcumin among various regions of brain at 2 h post-administration time point of single (100mg/kg b.wt) and repeated dose (100 mg/kg b.wt. for 28 days). The values are given as mean ± SD (n = 3), where ‘b’ – p < 0.01
CurQfen® Improves Memory and Learning in healthy rats – Behavior studies
The effect of brain delivery of curcuminoids was also studied on behavioural changes with a set of behavioural studies on vehicle control. In RAM test, CurQfen® treated animals had shown significant reduction in working memory error and reference memory error as compared to unformulated curcumin treated animals, which clearly shows the improvement on cognitive function upon CurQfen® administration.
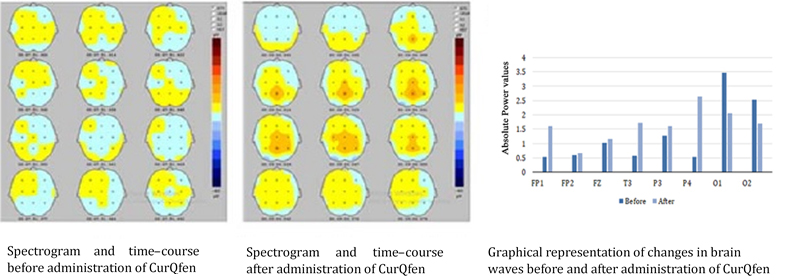
CurQfen® positively affects the Brain waves: The first line imaging evidence for BBB-permeability in human beings
CurQfen® was also investigated by Electroencephalography imaging (EEG) and showed for the first time that CurQfen®-curcumin can modulate brain waves in human volunteers as a proof of its BBB-permeability and brain penetration as compared to Unformulated curcumin which failed to make significant changes as observed with placebo (Nutritional Neuroscience, 2020).
.
What is CurQfen®?
A 100% natural green formulation of curcumin and fenugreek dietary soluble fibre clinically validated for enhanced bioavailability
Is CurQfen® organic and clean label?
CurQfen® is the first organic bioavailable curcumin in the market place. It contains only two GRAS-listed kitchen spices, namely curcumin from turmeric and dietary fibre from fenugreek manufactured using our water-based process of FENUMATTM. Both Organic and Non-organic grades are available.
is CurQfen® safe?
CurQfen® is absolutely safe. It has acute, repeated dose (28-days), subchronic (90 days) toxicity studies and genotoxicity studies (Food & Function, 2015). It has been extensively investigated for human safety, especially with respect to liver toxicity in the light of some recent support on hepatotoxicity issues with respect to some other curcumin supplements (Unpublished). No safety issues have been reported on CurQfen® and it is Self-affirmed GRAS.
Where is CurQfen® manufactured?
CurQfen® is manufactured by Akay Natural Ingredients Private Limited’s GMP certified manufacturing facility using patented natural technology FENUMAT™

